Cover image: AlexanderAlUS, licensed under CC BY 3.0.
Table of Contents
So here’s something pretty wild to think about. The graphene properties we’re about to dig into sound almost too good to be real. We’ve got a material that can basically “repair” itself and end up looking brand new, with no defects whatsoever. It has incredibly high electron mobility at room temperature, and it actually has the lowest known electrical resistance at room temperature — even lower than silver, which used to hold that crown. On top of all that, a single atomic layer of it absorbs roughly 2.3% of white light, which sounds like a small number until you realize we’re talking about something that’s literally one atom thick. Oh, and it’s also extremely thermally conductive, plus it has the highest mechanical strength ever measured — about 200 times stronger than steel.
Sounds too good to be true, right? But it’s real. The material is called graphene, and it’s been turning the materials science world upside down for the past two decades or so.
One Element, Totally Different Personalities
Here’s a question that sounds kind of crazy at first: can a single chemical element, in its pure state (so not mixed with anything else), be both the hardest thing you’ve ever seen and the softest? Can it be a conductor and an insulator at the same time? Can it even pull off something weirder — like dissipating heat in one direction while blocking heat in the other direction, all in the same piece of material?
Imagine you’re building a tube — or, more practically, a rocket nozzle, which is basically a tube anyway. You want that tube to carry heat along its length, but block heat from going through the wall. That sounds impossible. But it’s not.


Image credit: U.S. Army Materiel Command / CC BY-SA 2.0 (via Wikimedia Commons)
If you want to keep a rocket nozzle from melting or falling apart while all those super-hot exhaust gases blast through it, you make it from something called PyroCarbon. PyroCarbon does exactly the trick we just described — it pulls heat lengthwise toward the cooler parts of the rocket, while keeping heat from punching through the wall. And here’s the kicker: PyroCarbon is just pure carbon. Nothing fancy added. We’ve been making rocket nozzles out of this stuff since around the 1960s, when PyroCarbon was first discovered.
I’m not going to drag you through the full electronic structure of carbon atoms here, but there’s one thing worth mentioning. Carbon forms two main types of bonds. The first one (called sp3) gives you the diamond structure, where every atom is bonded to three neighboring atoms in a tetrahedral arrangement — meaning the bonds aren’t all flat in the same plane. The second type (sp2) also has each atom bonded to three neighbors, but this time everything sits in a single plane, kind of like a honeycomb. When you stack a bunch of those flat layers on top of each other, you get graphite.
And here’s what really matters: the bonds within each layer are way stronger than the bonds between the layers. That’s why adjacent layers in graphite slide past each other pretty easily, and can even peel apart. When that happens, you end up with thinner graphite crystals — but they’re roughly the same overall structure, just thinner, because the bonds inside each layer are still rock-solid.
But it’s not enough to just say “graphene is one layer of graphite.” Graphene is specifically a single layer of graphite that’s isolated enough from its surroundings that we can treat it as something on its own.
Why Nature Said “No” to Graphene for a Long Time
It’s pretty obvious in hindsight that researchers tried to get to graphene by thinning down graphite. Some early attempts go all the way back to 1960, when scientists managed to get down to about 15 layers. But despite tons of effort, nobody could get to a true single layer. After years of failure, people started to believe that standalone graphene basically couldn’t exist — that it wasn’t stable compared to curved carbon structures like soot, fullerenes, or nanotubes. The thinking was that any graphene you managed to make would just immediately curl up into one of those other shapes.
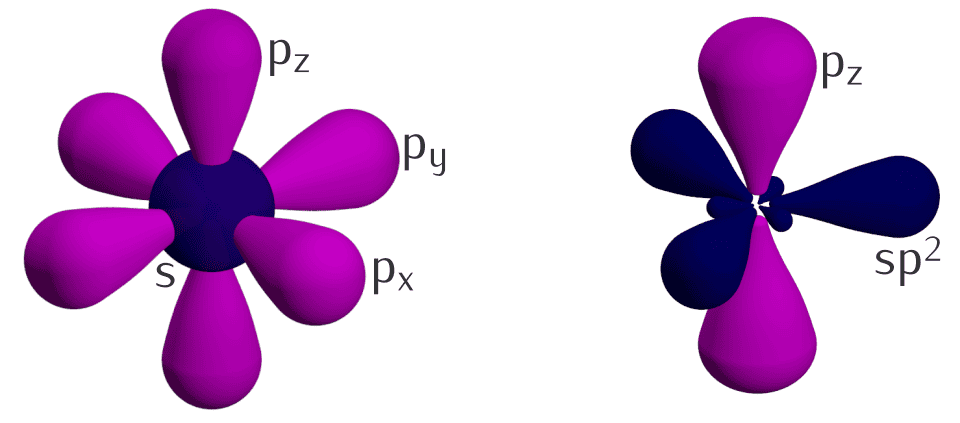
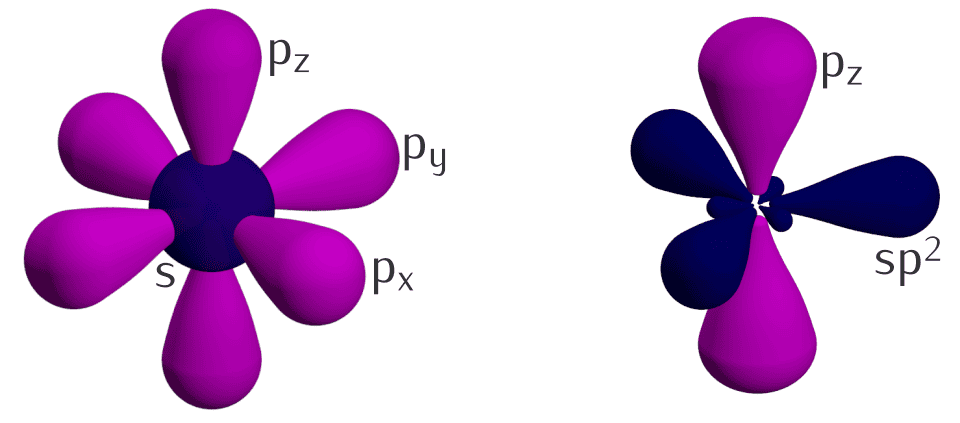
Image credit: Ponor / CC BY-SA 4.0 (via Wikimedia Commons)
The reasoning here isn’t crazy. Nature is pretty strict about not letting tiny crystals just grow on their own. Crystal growth needs high temperatures, and high temperatures mean thermal fluctuations, which mess up the stability of macroscopic 1D and 2D shapes. You can get flat molecules and nano-scale crystals, but as you make them wider, the thermal vibrations get more intense — and they’re different in different directions on a macro scale. So the 2D crystallites end up breaking down into a series of stable 3D structures instead.
But just because nature won’t let 2D crystals grow on their own doesn’t mean people can’t be sneaky about it. The trick is to grow a 2D monolayer inside or on the surface of another crystal — basically as a normal part of a 3D system — and then strip the host crystal away at a temperature low enough that the thermal fluctuations can’t tear the 2D layer apart.
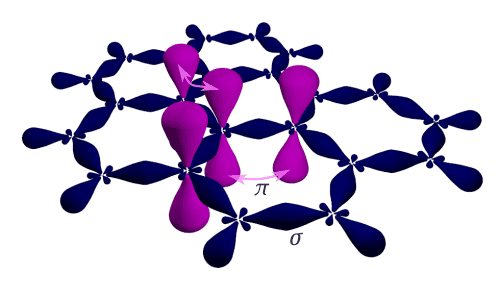

Image credit: Ponor / CC BY-SA 4.0 (via Wikimedia Commons)
The whole “is graphene stable or not” debate finally got settled in 2004. That’s when Konstantin Novoselov and Andre Geim, along with their colleagues, published papers showing they’d actually gotten “independent atomic crystals that are strictly two-dimensional (2D) and can be understood as individual atomic planes extracted from massive graphite crystals or as unwound single-layer nanotubes.” These were Russian researchers based in Manchester (or English researchers from Russia, depending on how you want to look at it), and they ended up winning the Nobel Prize for this discovery in 2010.
What’s funny is how simple their technique was. They took a graphite crystal and basically thinned it down using regular adhesive tape — peeling off layer after layer — and then transferred the super-thin graphite onto a specially prepared oxidized silicon wafer for testing.
People often call this the “adhesive tape technique” or “scotch tape method,” and even though it sounds almost too simple to be real, it actually produces crystals with great structural and electronic quality, sometimes reaching millimeter dimensions. The researchers showed that standalone graphene is stable under normal ambient conditions and is high quality to boot. Geim and Novoselov actually used the same technique to isolate 2D layers of several other layered compounds too, not just graphene.
The Carbon Family Tree
When we talk about graphene, it’s worth pointing out that it’s basically the ancestor of a whole family of carbon materials. At the far end of the family, you’ve got graphite — which is theoretically just an infinite stack of graphene layers — and PyroCarbon, which can be thought of as graphite with defects. But there are also nanomaterials in the family — fullerenes and nanotubes. Fullerenes are zero-dimensional (because they’re like little spheres that someone deliberately rounded off, so they don’t really extend in any direction), while nanotubes are two-dimensional.
Now here’s the part that really surprised everyone: it turned out that isolated 2D crystals weren’t just stable at room temperature and in normal air — they actually kept their macroscopic properties and high quality. The mobility of charge carriers stayed almost unchanged. This is super obvious in this material, where electrons can travel thousands of interatomic distances without bouncing off anything.
This discovery basically kicked off an avalanche of graphene research. Within just a few months, scientists had figured out how to do epitaxial growth of graphene layers on metal carbides (especially SiC) by sublimation, plus direct vapor deposition (the HDP process) on metals. For the metal substrate version, researchers also developed ways to transfer graphene from where it was grown onto different substrates with insulating layers. That made the whole thing actually usable for technology, not just a lab curiosity.
One question that came up a lot was: how many layers do you need before the structure stops being “2D”? For graphene specifically, this has been worked out pretty clearly. The electronic structure changes as you add layers, and by the time you hit 10 layers, you’re basically at the 3D boundary (graphite). Based on electronic structure, graphene with one layer, two layers, and a few more (3 to under 10) can be thought of as three different types of 2D graphene crystals. Anything thicker than that, you can pretty much treat as a thin film of graphite for any practical purpose.
The structure was studied using transmission electron microscopy (TEM) on carbon sheets suspended between metal lattice bars. The TEM showed the expected hexagonal lattice pattern. Interestingly though, the graphene planes aren’t perfectly flat. The suspended graphene actually showed bumps on the flat layer with an amplitude of around 1 nm. These might be unavoidable — possibly because of the inherent instability of 2D crystals — or they might come from impurities (adsorbates, basically) that show up in pretty much all TEM images of the material.
Here’s something cool: graphene can actually “fix” holes in its own layers, as long as there are carbon-containing molecules (hydrocarbons) around. And when you bombard the holes with carbon atoms, they get completely closed up — the new carbon atom slots right into the opening and matches up with the hexagonal pattern perfectly. Self-repair, basically.
Graphene Properties That Break the Rules of Physics
It has the lowest electrical resistance of any material we know about. It’s pretty different from most conventional 3D materials. On its own, this 2D crystal is a semimetal or zero-gap semiconductor. Way back in 1947, scientists figured out that the electrons and holes in this kind of structure have zero effective mass — yeah, you read that right, zero — which means they behave like relativistic particles described by the Dirac equation. That’s why these electrons and holes are called Dirac fermions, and the six corners of the Brillouin zone are called Dirac points. For more on the discoveries that shaped modern atomic physics, the early 20th century was truly a golden age.


Image credit: Ponor / CC BY-SA 4.0 (via Wikimedia Commons)
It has incredibly high electron mobility at room temperature — over 15,000 cm²V⁻¹s⁻¹. The mobility of holes is roughly the same. What’s interesting is that mobility is almost independent of temperature between 10 and 100 K. The mobility limit at room temperature comes out to about 200,000 cm²V⁻¹s⁻¹ at a carrier density of 10¹² cm⁻². That works out to a graphene layer resistance of around 10⁻⁶ Ω·cm — lower than silver, which previously had the lowest known resistance at room temperature. Now, when graphene sits on a SiO₂ substrate, electron scattering goes up, which drops the mobility limit to about 40,000 cm²V⁻¹s⁻¹. Still pretty incredible, just not record-breaking.
The optical properties are weird too. That single atomic layer absorbs around 2.3% of white light, which is unexpectedly high for something so thin. It comes from the unusually low-energy electronic structure of the graphene monolayer. The width of the graphene gap can be tuned anywhere from 0 to 0.25 eV (which corresponds to a wavelength of about 5 micrometers) by applying a voltage to part of a field-effect transistor. Graphene nano-traps can also be tuned in terahertz mode using a magnetic field. So basically, graphene is transparent — but not perfectly so. A single layer still blocks over 2% of light.
For thermal conductivity, this carbon material comes in somewhere between 4,800 and 5,300 Wm⁻¹K⁻¹ at temperatures close to room temperature, measured using a non-contact optical technique. Those values are actually higher than what’s been measured for nanotubes or even diamonds. One way to make sense of this is to think of graphite as a 3D version of graphene, where thermal conductivity within the layer plane goes over 1,000 Wm⁻¹K⁻¹ (which is comparable to diamond). In graphite, conductivity along the c-axis is roughly 100 times less because of the weak bonds between layers and the larger gap between layers.
Light as a Feather, Tough as Nothing Else
Graphene is the strongest material that’s ever been tested. Period. The breaking strength is 200 times higher than steel, and the tensile modulus (stiffness, basically) is 1 TPa. The catch is that pulling it cleanly out of graphite still needs some technological work before it’s cheap enough for industrial use. Besides being incredibly strong, it’s also super light — about 0.77 mg/m². Maybe this makes it clearer: a 1 m² graphene net weighing about as much as a single cat whisker could hold up a four-kilogram cat. Wild, right?
People have actually made graphene paper at this point. I’ll skip the prep details, but here’s how one of the inventors put it: “Not only is it lighter, stronger and more flexible than steel, but it can also be recycled, it is environmentally friendly and cost-effective to use…” And then: “Just as large aerospace companies are replacing metals with carbon fiber and other carbon materials, graphene paper will now replace them with its incomparable mechanical properties.”
Two-layer (or “bilayer”) graphene has some interesting electrical properties that make it a strong candidate for optoelectronic and nanoelectronic stuff. You usually find the bilayer version in one of two configurations — either twisted, where the two layers rotate relative to each other, or in Bernal stacking (named after Bernal, who figured out what graphite’s structure actually looks like), where half the atoms in one layer sit directly on top of half the atoms in the other. The way the layers stack and orient strongly affects both the optical and electronic properties. One way to make bilayer graphene is using HDP, which can produce large bilayer surfaces that are pretty much all in Bernal geometry.
Where Graphene Is Actually Showing Up
Out of all the potential uses for this material, several are already in development. We’re talking about light, thin, flexible, but durable screens, electronic circuits, solar batteries, and various medical, chemical, and industrial processes that get improved or made possible by carbon-based materials.
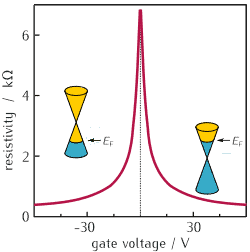

Image credit: Ponor / CC BY-SA 4.0 (via Wikimedia Commons)
For integrated circuits, it basically checks all the boxes: high charge carrier mobility, low noise, which makes it great for use as a channel in field-effect transistors. The hard part is producing single layers of graphene — and it’s even harder to make them on top of a suitable substrate. Researchers are working on methods to transfer single graphene layers directly from where they form (whether from mechanical thinning or graphitization of an SiC surface) onto whatever surface you actually want to use. The smallest transistor ever made, as of the relevant work — one atom thick and 10 atoms wide — was built in 2008, and a graphene integrated circuit followed just a month later. By June 2011, there was an announcement that the first graphene-based integrated circuit had been developed. This thing operated up to 10 GHz at temperatures as high as 127°C. Combined with developments in quantum computing, graphene-based circuits could play a major role in the next generation of processors. Then in November 2011, researchers showed that ink-jet printing could actually be used to make graphene devices. Pretty cool.
The combination of high electrical conductivity and good optical transparency makes graphene a serious candidate for transparent conductive electrodes — stuff like touch screens, liquid crystal displays, organic photovoltaic cells, and organic LEDs. Mechanical strength and flexibility give graphene a real edge over indium tin oxide, which is brittle. Plus, graphene films can be deposited from solution onto large surfaces. The HDP technique has produced continuous, breathable films with large surface area and high conductivity for photovoltaic devices. People have also shown organic LEDs with graphene anodes. And the electronic and optical performance of graphene-based devices is comparable to what you’d get with indium tin oxide.

Image credit: Ponor / CC BY-SA 4.0 (via Wikimedia Commons)
Here’s a fun one. Graphene oxide membranes let water vapor pass through, but they’re impermeable to other liquids and gases — even helium, which is notoriously sneaky and gets through almost everything. Researchers actually used this property to further distill vodka, getting higher alcohol concentrations. And they did this at room temperature, with no heat or vacuum, which is otherwise pretty standard for distillation. If this kind of membrane gets developed and commercialized further, it could basically revolutionize biofuel production and the alcohol industry.
Back in 2008, there was an announcement that highly transparent graphene films had been produced using HDP on a large scale. The researchers made ultra-thin graphene layers by first depositing carbon atoms from methane onto a nickel plate, forming graphene films. Then they put a protective thermoplastic layer on top of the graphene and dissolved the nickel underneath using acid. In the final step, the ultra-thin graphene (still protected by plastic) gets attached to a flexible polymer layer, which can then be built into an OPV cell — that’s organic photovoltaic, basically a graphene solar cell. Graphene polymer sheets up to 150 cm² have been produced, and you can get dense arrays of flexible OPV cells out of them. Eventually, a printing process can be applied, which means large areas covered with cheap solar cells, kind of like how newspapers get printed at huge scale.
A Sensor That Notices Single Molecules
In theory, it’s a phenomenal sensor because of its 2D structure. The fact that its entire volume is exposed to the environment makes it really good at picking up adsorbed molecules. But there’s a catch — similar to nanotubes, graphene doesn’t have “dangling” bonds on its surface, so gas molecules can’t just stick to it easily. So pure graphene by itself isn’t actually that sensitive. The sensitivity of graphene chemical gas sensors can be cranked up dramatically by adding functional groups — for example, by coating the graphene with a thin layer of some polymer.
The thin polymer layer acts kind of like a “concentrator” that absorbs the gas molecules. When molecules get absorbed, they cause local changes in the electrical resistance of the graphene sensor. This effect exists in other materials too, but graphene is way better because of its high electrical conductivity (even when only a few carriers are around) and low noise, which means even tiny resistance changes can be picked up.
Graphene nanoribbons are basically individual graphene layers cut along specific patterns (zigzag, armchair) to get specific electrical properties. Calculations predict that zigzag ribbons should always be metallic, while armchair ones can be either metallic or semiconducting depending on their width. But calculations also show that armchair nanoribbons are semiconductors with an energy gap that grows as the ribbon gets narrower — and experimental results back this up.
Zigzag ribbons are also semiconductors, and they show spin-polarized edges. Their 2D structure, high electrical and thermal conductivity, and low noise make nanoribbons a possible alternative to copper for integrated connections between components. Large quantities of width-controlled nanoribbons can be made using the nanotomy process.
There’s a lot about graphene that’s just plain amazing. Completely unexpected stability for a flat 2D structure. Retention of macroscopic properties and high quality, with charge carrier mobility staying almost unchanged. Graphene electrons can travel thousands of interatomic distances without getting deflected.
Just to recap a few applications that have moved past “potential” and into reality: the smallest transistor ever made; transparent conductive electrodes (touch screens, etc.); graphene oxide membranes that block gases and vapors (even helium) but let water vapor through; large areas covered with cheap solar cells starting to come into use; and detection of individual molecules.
What’s left to say? Carbon is an element that just doesn’t stop surprising us.
References:
- Geim, A. K.; Novoselov, K. S. (26 February 2007). “The rise of graphene”. Nature Materials.
- Peres, N. M. R.; Ribeiro, R. M. (2009). “Focus on Graphene”. New Journal of Physics.
- Zhu, Shou-En; Yuan, Shengjun; Janssen, G. C. A. M. (1 October 2014). “Optical transmittance of multilayer graphene”.
- Lee, Changgu (2008). “Measurement of the Elastic Properties and Intrinsic Strength of Monolayer Graphene”.
- Joshi, Rita (8 April 2024). “Can Graphene Be Mass Produced?”
- Brodie, B. C. (1859). “On the Atomic Weight of Graphite”. Philosophical Transactions of the Royal Society of London.
- Meyer, J.; Geim, A. K.; Katsnelson, M. I.; Novoselov, K. S.; Booth, T. J.; Roth, S. (2007). “The structure of suspended graphene sheets”. Nature. 446