Cover image: Ministry of Information Photo Division Photographer, public domain
Table of Contents
It is probably the most famous speck of dust in history. No one knows where it came from, although there have been many guesses. But it was almost as if a benevolent God pursed His lips and blew – sending it wafting in the summer air, through the laboratory window, and onto the petri dish that was sitting before researcher Alexander Fleming. It was a speck of dust that would create a revolution in medicine and it held a secret that would save the lives of millions of people in the years to come. That secret was Alexander Fleming discovery of penicillin — one of the most remarkable accidents in the history of science. Alexander Fleming was just the right man to be sitting there that day when the unexpected dust appeared. He was a man of exceptional skill, patience, and perseverance – all the qualities that would be needed to solve the mystery that rose before him.
Early Life and Childhood in Scotland

Image credit: Imperial War Museums, public domain
Fleming was a Scotsman. He was born August 6, 1881, in Ayrshire, the youngest of eight children. Alexander’s father was a farmer and he hoped from the beginning that his youngest son would be the one to carry on his work on the family farm. Like the parents of many great people, he would have to set his own dreams aside so his child could pursue his unique talents and interests.
At first, Fleming considered the idea of becoming a farmer. Like most boys, he enjoyed the country life. He helped his father cut peat; he fished for trout in neighboring streams; he snared rabbits with his bare hands, and he roamed free in the countryside with his father’s sheep. He once recalled: “Living as we did at the end of the road, and upon the edge of the moor, we considered ourselves a bit better than the boys in the town; they did not know how to climb and they did not know where to find peewits’ eggs…I might have stayed on the land and become a farmer. I might have been a very good farmer, who knows, and I might have had the finest herd in the country.” He might have but he didn’t. Instead, what he reaped from the farm was chiefly a deep understanding and respect for nature and acute powers of observation.
Education and Career at St. Mary’s Hospital
Fleming had some early schooling and then, when he was almost fourteen, his life turned in an entirely new direction. He was sent to London to live with his older brother Tom, who was an ophthalmologist. His parents valued education and knew he would learn a great deal more in London than in the country schools of Scotland. What they didn’t expect was that Fleming, once exposed to city life, would never again return to the farm. He had a Scottish accent his entire life, but from the time he first saw London, Alexander Fleming adopted England as his home.
The year now was 1895 – the height of Queen Victoria’s reign. She had just given birth that year to her new son, the future King George VI. The underground subway, powered by steam, rumbled beneath London’s busy streets; Oscar Wilde was in court defending himself against attacks on his homosexuality; H.G. Wells released his astounding book The Time Machine; the foundation stone for Westminster Cathedral was laid, and across the channel in France, the great medical scientist Louis Pasteur had just died. The next man to contribute as much to medicine as Pasteur did would be Fleming himself.
At the time, he was attending the same boy’s academy where the poet Robert Burns and novelist Robert Louis Stevenson had been educated. In his free time, he spent with his brothers. By now four Fleming brothers were living in one house on Marylebone Road, with their sister Mary serving as a housekeeper. They explored the city of London together on horse-drawn buses.
For a short time Fleming worked as an office clerk for a shipping firm, a job he didn’t enjoy, but which he later admitted gave him some practical skills and the attention to detail that later helped him in science. He also joined a sports group called the Scottish Volunteers and became one of their best swimmers. His interest in swimming was pivotal – in an indirect way it would lead to his career.
When Fleming was twenty he came into an inheritance from an uncle and had to decide what to do with the funds. His brothers suggested he study medicine as they had; one was a general practitioner and two others were in ophthalmology. His brother Thomas was so adamant about this idea, that Fleming relented. The problem was that there were twelve medical schools in London, all under the jurisdiction of the University of London, and Fleming had no idea which one to choose. Then he recalled that he once played water polo against one of the twelve St Mary’s schools. That seemed as good a reason as any to choose a school so in 1902, aided further by a scholarship, he enrolled.

Image credit: Vera de Kok, / CC BY-SA 4.0 (via Wikimedia Commons)
What Alexander Fleming found at St. Mary’s that turned out to be far more important than water polo, was an eminent bacteriologist named Sir Almoth Wright. Wright was a brilliant Irishman, one of the first physicians to use the typhoid vaccine on his patients. He was also a cultured man and an aristocrat – his friends included George Bernard Shaw, who used Wright as the model for his hero in the play The Doctor’s Dilemma. Wright’s specialty, and a subject he often lectured on at St. Mary’s, was the immunology.
While Fleming studied under Wright, he learned to observe closely, to develop a detailed memory, and to work economically. The last, he said, came easily, for he described himself as: “just a canny Scot who cannot bear to throw away anything – not even a contaminated Petri dish.” Wright was an involved professor – he worked closely with his students and was always accessible to them. A French visitor to the school once reported how astounded he was to see the student Fleming walk up to the professor while he was lecturing and, without a word, prick his finger for a blood sample he needed. Wright continued talking without paying the least attention, and Fleming, looking very serious, hustled his sample back to his worktable.
Fleming became a hard worker and was such an outstanding student that he took all the academic awards when he graduated in 1908, at the age of 27. These included awards and scholarships in Physiology, Hygiene, Medicine, Pathology, and Pharmacology. He also passed his surgical exams with honor and became a Fellow of the Royal College of Surgeons. Surgery, however, didn’t interest him. Under the influence of Wright, he’d become much more interested in the tiny little creatures that could only be seen through a microscope, which today we call germs, which back then were called “microbes.” After graduation, Fleming joined the Inoculation Department at St. Mary’s, which was under Wright’s directorship, and began to pursue his own research.
Fleming worked well under Wright even though the men had a basic difference of opinion about what the focus of that work should be. Wright’s main interest was immunology – the science of making people immune to disease, usually through vaccination. He wasn’t a big fan of what is called chemotherapy – the use of drugs to treat disease. Later, Wright would worry about the time and faith Fleming poured into the drug penicillin. But he always remained proud of his student – proud enough to alert the London Times to the news that the glory for the discovery of penicillin belonged to Fleming. This made two other men who had worked on penicillin after Fleming very distressed, as science, like most fields, has a fair amount of rivalry and competition.
The laboratory in which Fleming worked at St. Mary’s was under Wright’s careful direction for over 45 years. While Fleming was there he won award after award for his research and discoveries, still while he was in his late 20’s. Soon he was teaching his own classes. After classes, he would retreat to his lab, where he often worked as late as 2 am. Students from the university would sometimes drop by and share a beer with him.
Image credit: Cacycle, public domain
Fleming was a short, handsome man with bright blue eyes, and he always worked with a cigarette dangling out of his mouth. When he was a child, he once collided with another boy while running around the corner of a wall and smashed his nose. The accident left the upper half of his nose flattened – like a boxer’s nose – and for the rest of his life, it was his most unique feature. He was a very taciturn man, given to few words. But Ernst Chain, who later shared a Nobel Prize with him, said that despite his quietness, he gave the impression of being a warmhearted person, even though, in his words, “he did everything to appear unemotional and aloof.” Aloofness aside, Fleming was always a favorite with his students.
Even with his tremendous workload, Fleming always found time for sports. The laboratory had an excellent shooting team and he was one of their crack shots. Once, when lecturing a group of young medical students, he promoted the idea of sports saying: “You should know even at this stage of your career that there is far more to medicine than mere book work. You have to know men and you have to know human nature. There is no better way to learn about human nature than by indulging in sports, more especially in team sports.”
World War I: From Laboratory to Battlefield
The sports and the studying came to an abrupt halt in 1914 when World War I began. For the next few years, Fleming’s work would be conducted, not in sterile, orderly laboratories, but in the crowded, gruesome hospitals of war torn Europe. He would play the role not of the detached, curious scientist, but of the impassioned doctor, filled with anguish at the sight of enormous human suffering that he could do little to relieve.
Shortly after his thirty-third birthday Fleming became an officer in the Royal Army Medical Corps and headed out to France, the scene of some of the war’s heaviest fighting. He was stationed in Boulogne, at the city’s casino, which had been hastily converted into a hospital. The casino was packed with badly wounded soldiers, many of them suffering from gangrene. Their moans and screams of pain filled the air day and night. Fleming described the improvised laboratories at the casino as “two subterranean bathrooms, which were periodically flooded with sewage.” He was shocked by what he considered the unnecessary fatalities – men dying of wounds that should have healed, antiseptics that failed to do what they were expected to do, bacteria that went unchecked no matter what the doctors tried. Only a short time after his arrival he began to experience a sense of failure as a doctor, as a man who allegedly was a healer.
In 1915, Fleming married an Irish army nurse while he was on leave in Britain. Her name was Sarah McElroy but her friends knew her as Sareen. Sareen’s sister had also married Fleming’s older brother, John. Fleming was devoted to his wife and when she died 34 years later, in 1949, he suffered a long, intense period of grief.
After he married, Fleming returned to France to resume his sad and frustrating duties in the war hospital. The more he encountered the deep and jagged wounds from artillery explosions, the more he was convinced that dead tissue was a culture medium for infection. He began to advise surgeons to cut away the dead tissue, a process now called debridement. He also began to re-evaluate the work of the famous 19th century English surgeon Joseph Lister. Lister, who was a good friend of Louis Pasteur, was the first to push for more sanitary and sterile conditions during surgery, including the cleaning of wounds with antiseptics. But Fleming found that, although Lister’s suggestions were very valuable, the use of antiseptics in wounds that were very deep, actually promoted the growth of bacteria – especially gas gangrene. He began using and recommending saline solutions instead. Even though his later discovery of penicillin became the chief work for which he was remembered, Fleming’s findings with antiseptics are considered by many physicians to rank among his greatest work.
Wounds and injuries were enough to keep doctors busy during the war but then another medical crisis hit – the terrible flu epidemic of 1918, which soon spread throughout the world. At the time, no one knew what caused the illness. It would be fifteen years before the influenza virus was isolated and identified. As the flu swept through the British army Fleming, like other doctors, had no choice but to watch as soldiers either recovered on their own or died of pneumonia. Trying to give some aid to the victims of flu, and trying to save the limbs and lives of soldiers with gangrene, were occupations that kept Fleming busy, exhausted, and discouraged throughout the war. When the war was over he was 37 years old, he was a captain. He had frequently been cited for his achievements, yet he was deeply disheartened. He returned to London with a mission.
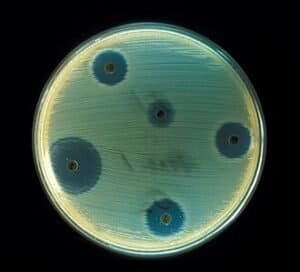
Image credit: CDC / Provider: Don Stalons, public domain
Once back home, Fleming decided to devote himself to immunology in the hopes he could find some method for effectively destroying bacteria without harming healthy tissue. His experience and experiments had convinced him that the best weapons against bacteria were the body’s own cells. He said: “I could never forget the importance of the body’s natural defenses.”
Fleming’s first major work after the war was with an enzyme called lysozyme, which turned out to be a precursor of penicillin. He found lysozyme in certain body fluids, such as tears and saliva. He discovered when he added it to a petri dish that it actually dissolved some of the bacteria in the dish. Although the bacteria destroyed wasn’t virulent, or harmful, it made him hopeful that he might find a stronger substance that could destroy harmful bacteria. His paper on his experiments with lysozyme was one of his most important works. It bore the title: Observations on a Remarkable Bacteriolytic Substance Found in Secretions and Tissues and gained attention throughout the scientific community. Only a few years later, while pursuing this lead, he would stumble across another discovery, one of the greatest in the history of medicine.
Alexander Fleming Discovery of Penicillin in 1928
It happened in 1928 when Fleming, now known simply as Flem, was a gray-haired man of 47. He was a meticulous, even finicky researcher and he was known for his unusual habit of never throwing anything away. That included his cultures, which he often saved for a long time. On this summer day, he would be very grateful that he did.
Fleming was experimenting with a variety of compounds when suddenly he noticed a strange phenomenon in one of his Petri dishes. A petri dish is a shallow glass container filled with a medium on which colonies of bacteria are grown for study. In this particular dish, he’d been growing very virulent Staphylococcus germs, for a chapter in a book he was writing. Now he saw that mold was growing on part of it. He didn’t know what had settled onto the dish to cause the mold but legend has it that a speck of dust with mold spore in it had floated in from a beer barrel in a nearby saloon. The dust settled on the petri dish, the mold grew, and to Fleming’s astonishment, something else happened. The yellow staphylococci colonies become transparent.
Years later, when Fleming was touring a very modern and sterile research laboratory that was air-conditioned and completely dust-free, his guide said: “What a pity you didn’t have such a place as this to work in. Who can tell what you might have discovered?” “Not penicillin,” said Fleming with a smile.
When Fleming first saw the mold growing in his contaminated petri dish and the disappearance of the germs beneath it, he wasn’t sure what to think. “That’s funny,” he said to a colleague as he studied the dish. It reminded him of what had happened in his experiments with lysozyme – some unknown agent in the mold had killed what had once been living germs. When a visiting Rhodes scholar wandered in a little while later, Fleming took him to the dish and said: “You think yourself a clever fellow; explain this one.”

Image credit: Science Museum London, CC BY-SA 4.0 (via Wikimedia Commons)
Fleming examined the dish more closely under his microscope and saw the mold in action – he could actually see the bacteria shrinking. Still, he had no idea of the significance of what he’d discovered. He wrote later: “Nothing is more certain that, when I saw the bacteria fading away, I had no suspicion that I had got a clue to the most powerful therapeutic substance yet used to defeat bacterial infections in the human body.”
Fleming identified the greenish mold that killed the bacteria as Penicillium, or “little brush,” – because of the brush like ends of the minute fibers on the mold. Then he named the liquid that came from the mold penicillin – a name that would soon go down in history. Penicillin was, and is, an antibiotic – a living substance that fights other living substances that are dangerous.
For hundreds of years, in primitive societies all over the globe, it was known that molds had some healing value. In some cultures, they made poultices of different molds and fungi to apply to open wounds. But before Fleming, scientists had never isolated the agents that had the beneficial effect, and in fact, most scientists had never tried. They paid little attention to the cultural use of molds in healing, which they considered superstition and folklore.
Fleming obtained a pure culture from his sample and began to grow it on stale bread, cheese, and fruit, but he soon found it flourished best in meat juices. With these small amounts, he continued his experiments and found that over and over again he could prove its potency, even when it was diluted. Following the example of Louis Pasteur, he decided to experiment on mice. He first injected them with dangerous germs like staphylococci, streptococci, and pneumococci – the germs that cause staph and strep infections and pneumonia. Then he inoculated them with his new penicillin liquid. Not only did the germs disappear, but the mice suffered no ill effects. Penicillin seemed to be non-toxic. He kept testing on more mice, and then on rabbits. He discovered his liquid mold prevented the growth of germs even when it was diluted 800 times. When he finally published his findings the next year, in the British Journal of Experimental Pathology, he made what had to be one of the most modest understatements in medical history. He said: “Penicillin may be an efficient antiseptic for injection into areas infested with penicillin-sensitive microbes.” In fact, penicillin was about to become one of the world’s first miracle drugs.
First, however, there was a problem to be solved. Penicillin was very difficult to produce, except in small amounts. Fleming hired chemists to help him find ways to produce it in large amounts, but the mold was unstable and seemed to disappear right before their eyes. Eventually, he became discouraged. Although he continued to cultivate his molds, for the next ten years he did very little with them and began to accept the idea that there could never be any large-scale use of penicillin.
From Discovery to Life-Saving Drug: Florey and Chain
Alexander Fleming had written the first chapters on penicillin. Now it would be up to other scientists to finish the book. The two men who were most responsible for proving the therapeutic benefits of penicillin, and for starting its large scale production were the researchers Howard Florey, an Englishman, and Ernst Chain, a German Jew who had fled his homeland when Hitler came to power. He left behind a mother and sister, who were later killed in the concentration camps.
Fleming’s paper on penicillin had made a big impression on Florey and Chain, who were working together at Oxford at the time. For months they tried to extract the essential substance from Fleming’s mold, which Fleming had gladly contributed to the cause of further research. Finally, they obtained about a teaspoonful of yellow-brown powder – the first salt of penicillin. Florey and Chain began to produce first small, then larger amounts of penicillin in hopes they could create enough to conduct experiments, and find a way to mass-produce it. They proved its effectiveness, as Fleming had, on mice. Then in 1941, they gave the first dose of penicillin to a human being – a London policeman badly infected with staphylococci. The man was critically ill; his body was covered with abscesses, one of his infected eyes had been surgically removed, and he had a fever of 105 degrees. Almost every viable drug had been tried and none had any effect on him. Yet after a single injection of penicillin, his temperature dropped dramatically. He continued to steadily improve until the fifth day when the small supply of penicillin had been exhausted. Without it, he relapsed and died within a month.
After that, the researchers decided to limit their treatment to children, who required smaller doses of the drug. A fifteen-year-old boy, dangerously infected with streptococci, was injected with penicillin and overnight was rescued from death. A few months later Fleming himself injected penicillin into the spinal canal of a dying friend who then recovered within a week.
Meanwhile, events were happening that made them work with penicillin even more imperative and which gave researchers the support they would need to finish it. World War II had begun. The allies, anxious to find new and better ways to treat wounded soldiers and to prevent the diseases that decimated troops in World War I, were desperate for the new drug. But England, crippled by bombings, was unable to provide the resources necessary to continue the work. Even Florey’s own home had been bombed and he’d narrowly escaped death. On a secret flight in which he carried penicillin extract wrapped in cotton wool, Foley came to America where he hoped he could find the skills and resources that were needed to mass-produce the drug. He did. It was a long process of experimentation that involved several brilliant minds and the energy of countless workers, but finally, pharmaceutical companies in the United States began to produce huge quantities of penicillin to be shipped overseas and to be used at home. In 1942 there was barely enough penicillin to treat a hundred patients – a year later countless thousands of lives were being saved by the most powerful and least toxic of all antibiotics. On D Day, June 6, 1944, the ships that landed on the shores of Normandy were well supplied with thousands of ampules of purified penicillin.
Nobel Prize and Fleming’s Legacy in Medicine
In 1944 Fleming was knighted for his work with penicillin and in 1945, as the war was ending, he was awarded the Nobel Prize in medicine, a prize he shared with Howard Florey and Ernst Chain. In his humble speech to the Nobel committee, Fleming said his discovery rose “simply from a fortunate occurrence which happened when I was working on a purely academic bacteriological problem which had nothing to do with bacteriological antagonism, or molds, or antiseptics, or antibiotics. In my first publication I might have claimed that I came to the conclusion, as a result of serious study of the literature, and deep thought, that valuable antibacterial substance was made by molds, and that I set out to investigate the problem. That would have been untrue, and I preferred to tell the truth: that penicillin started as a chance observation. My only merit is that I did not neglect the observation, and I pursued the subject as a bacteriologist.” After he won the Nobel Prize, Fleming became the director of the Wright Institute of St. Mary’s, where he’d once trained and taught under Wright himself.

Image credit: Unknown author, public domain
Inspired by the success of penicillin, scientists went on to develop other molds that could fight disease, many of them derived from various soils. From the earth in New Jersey came streptomycin; from the earth in Venezuela came a mold that stopped an epidemic of typhus. Then came Aureomycin, from Missouri mud, and Terramycin from decayed Indiana soil. A new era of medicine had arrived, ushered in by the discovery of Alexander Fleming – the era of antibiotics. The effects of antibiotics were immediate and profound. Mastoid operations, which had been the fear of almost every parent of small children, almost disappeared. Now the dreaded ear infection could be treated with drugs instead. Within only a few years the number of mastoid operations in one Boston hospital declined from an average of 300 a year to only five. Appendicitis, peritonitis, meningitis, strep throat, and the often fatal illness known as “childhood fever” all lost their power to strike terror in the public. Newer, more powerful antibiotics were developed almost every year until there was a complete family of drugs effective against harmful bacteria. Today there are continuing refinements of those drugs, attempts to check allergies to them, and to modify the sometimes-excessive power of antibiotics. There is a growing concern too, that overuse of the drugs has weakened our natural immune systems. But it cannot be argued that penicillin, and the antibiotics that followed it, saved the lives of countless numbers of children and adults throughout the world.
Alexander Fleming enjoyed the exposure his success had given him. He began to come out of his shell and overcame his shyness to become a confident and relaxed speaker. He retired at the age of 67, saying that he felt the younger generation should carry on his work. Then he started a new hobby – one that combined his love of bacteriology with his interest in art. He painted bright abstracts of enlarged germ cultures and decorated his walls with them. He also continued to tinker in his laboratory, reluctant to completely give up his life’s work. On his 70th birthday, he said: “I have still got a few useful years before me. The happy man keeps working.” He was working in his laboratory the day before he died of a heart attack, on March 11, 1955. He died at his London home, only a few blocks away. In his laboratory, sealed in a special container, was the original mold – the one that grew from a mysterious speck of dust one day twenty-seven years before. Fleming’s ashes were placed in a crypt in St. Paul’s Cathedral.
It took the work of three leading scientists to eventually bring penicillin into mass production and use. But it was the experiments of Alexander Fleming that started the process and brought the potential of penicillin to the attention of the medical community. Probably no other drug in history has saved so many lives as penicillin. It was one of the first in a series of “wonder drugs” that dramatically influenced the quality of human life and lengthened our life spans. Fleming had always modestly called his discovery “a triumph of an accident.” In truth it may have started with an accident – a little speck of dust drifting in from some unknown place at just the right time – but he followed that accident up with keen observation and attention, intensive research, experimentation, and a willingness to seek the help of experts. It started as an accident. But it was Alexander Fleming who transformed that accident into a major discovery, and one of the greatest gifts to modern humanity.
References:
- The Cambridge Illustrated History of Medicine. Cambridge, England: Cambridge University Press, 1996;
- “Sir Alexander Fleming – Biography”. Nobel Foundation. Retrieved 25 October 2011.
- Penicillin Man: Alexander Fleming and the Antibiotic Revolution, Stroud, Sutton, 2004. Brown, Kevin;
- Fleming, Discoverer of Penicillin, Ludovici, Laurence J., 1952
- The Life Of Sir Alexander Fleming, Jonathan Cape, 1959. Maurois, André.
- Bennett, Joan W.; Chung, King-Thom (2001). “Alexander Fleming and the discovery of penicillin”. Advances in Applied Microbiology.
- Bickel, Lennard (1995) [1972]. Florey: The Man Who Made Penicillin. Melbourne University Press Australian Lives. Carlton, Victoria: Melbourne University Press
