Cover image: Marcin Białek, licensed under CC BY 3.0.
Table of Contents
In the 20th century, our whole picture of the universe got turned upside down. Scientists cracked the laws of the subatomic world, discovered a host of new elementary particles, and gradually came to realize that matter possessed fundamental properties nobody had even thought to look for. One of the most surprising of those properties was electron spin — the intrinsic angular momentum carried by every electron, independent of its motion. Its discovery didn’t come from a grand theoretical breakthrough. It came from a single experimental result that refused to fit any existing theory: the Stern-Gerlach experiment. What began as a relatively routine test of atomic structure ended up revealing something that would reshape quantum physics entirely.
The Stern-Gerlach Experiment: Setup and Predictions
In 1921, a physicist named Otto Stern conceived a straightforward idea. Take a beam of atoms, each with a single outer electron, and direct them through a non-uniform magnetic field. The magnetic field would exert a force on the atoms depending on how their magnetic moments were oriented, splitting the beam into separate streams. The setup was elegant, and the logic was sound. Nobody walked into that experiment thinking they were about to uncover something fundamental about the nature of matter itself.
At the time, there were three competing predictions about what the experiment would show, each rooted in a different theoretical framework.
The old Bohr theory held that atoms had no preferred orientation in a magnetic field — their magnetic moments should be distributed evenly in all directions. If that was correct, the magnetic field would scatter them smoothly and continuously, producing a smeared, diffuse pattern on the detector screen rather than any distinct, separated beams.
The Bohr-Sommerfeld theory offered a different prediction. It stated that the electron’s magnetic moment could only point in certain specific, quantized directions relative to the external field. For an electron in the so-called s state, this theory predicted the beam would split into three distinct streams: one central beam and one on each side, symmetrically distributed. The key requirement was that the number of resulting beams had to be odd.
Then there was the newer quantum framework developed by Schrödinger and Heisenberg, which pointed in yet another direction entirely. According to this theory, an electron in the s state carries no orbital magnetic moment whatsoever. If that was right, the non-uniform magnetic field shouldn’t produce any meaningful splitting at all.
Three theories. Three different predictions. The experiment would settle the matter.
Experimental Results: Two Beams Instead of Three
The result left the physics community genuinely puzzled.
The beam did split — but not into three streams. It is split into exactly two. And crucially, there was nothing in the center. The atoms landed on the detector screen in two distinct, well-separated clusters: one deflected in one direction, one deflected in the other, with a conspicuous gap in between. The outcome didn’t match any of the three predictions cleanly. The continuous scattering expected by the old Bohr theory was nowhere to be seen. The three-beam prediction of Bohr-Sommerfeld was clearly wrong. And yet something magnetic was definitely happening, which ruled out the “no moment” prediction from Schrödinger and Heisenberg as well.
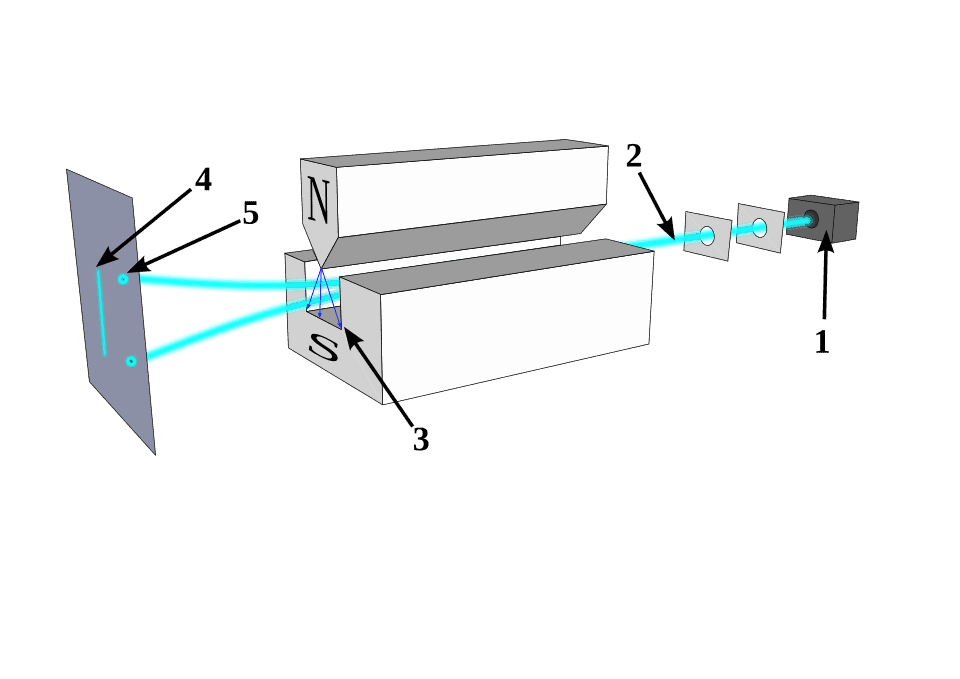
Image credit: Tatoute / CC BY-SA 4.0 (via Wikimedia Commons)
Two beams. An even number. That detail was more significant than it might appear, because all existing theories of angular momentum quantization predicted an odd number of possible orientations. Two beams implied a half-integer quantum number — something the theoretical framework of the time had no room for.
For several years, the result sat without a satisfying explanation.
Uhlenbeck and Goudsmit: The Path to Electron Spin
Fast forward to the summer of 1925. Two young Dutch physicists — George Uhlenbeck and Samuel Goudsmit — began an intensive collaboration at the University of Leiden. They made a complementary pair. Uhlenbeck was a rigorous theoretical physicist with a strong command of the emerging quantum formalism. Goudsmit had a different kind of talent — an almost detective-like ability to look at a collection of seemingly unrelated experimental data and identify hidden regularities. His colleague Isidore Rabi once remarked that Goudsmit thought like a proper detective, which, as it turned out, wasn’t purely a figure of speech. Goudsmit had actually attended a detective course the year before. Whether Rabi was aware of this remains unknown.
That summer, one of the problems they kept returning to was the anomalous Zeeman effect — a long-standing puzzle in atomic spectroscopy. The effect referred to the way certain spectral lines of atoms split into unexpected patterns when placed in a magnetic field, in ways that existing theory couldn’t fully account for. Physicists had been wrestling with it for years without a clean resolution.
Goudsmit was increasingly convinced that a simpler underlying picture had to exist. He had noticed that the mathematics worked out considerably better if the electron’s quantum number were allowed to take on half-integer values. This idea had already been touched on by Wolfgang Pauli earlier that same year, when he proposed a mysterious fourth quantum number — one that could only take half-integer values — and assigned it directly to the electron rather than to the atom as a whole. Pauli’s move had been a bold one, and it gave physics the famous exclusion principle. But Pauli was deliberately cautious about assigning any physical meaning to this new quantum number. He introduced it as a mathematical necessity and left it at that.
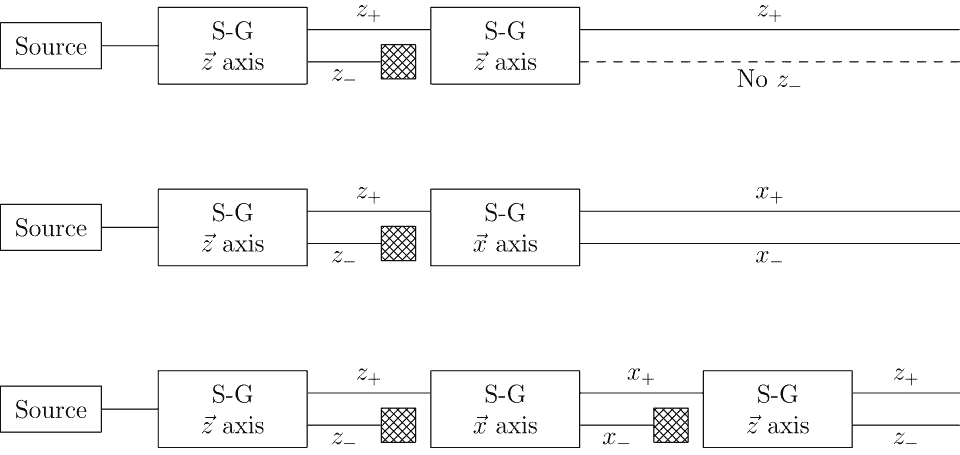
Image credit: Francesco Versaci, public domain
The Electron Spin Hypothesis: A New Quantum Property
This is where Uhlenbeck and Goudsmit made their decisive step.
Goudsmit began to wonder whether Pauli’s fourth quantum number might correspond to something physically real — specifically, whether the electron might be literally spinning around its own axis, the way a planet rotates as it orbits the sun. The analogy was imperfect, but the physical reasoning had a certain elegance to it.
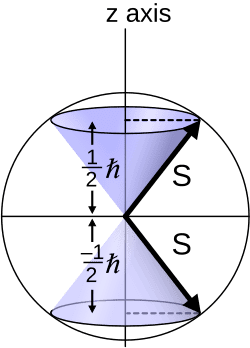
Image credit: Theresa knott / CC BY-SA 4.0 (via Wikimedia Commons)
A charged sphere rotating around its own axis is, in effect, a tiny loop of moving electric charge. Any such loop generates a magnetic dipole moment — it behaves, in other words, like a small magnet. If the electron possessed this kind of intrinsic rotation, it would carry its own magnetic moment entirely independent of its orbital motion around the nucleus. This internal magnetic moment — what came to be called spin — could then interact with the magnetic moments of other electrons in the atom, shifting their energy levels by small amounts and producing the complex fine structure observed in atomic spectra.
The quantitative picture supported the idea. If the electron’s spin quantum number had a value of one-half, the gyromagnetic ratio — a measure of how strongly a magnetic moment responds to an external field, commonly called the g-factor — worked out to approximately 2. That value matched experimental observations far better than the classical prediction of 1, which had long been a source of frustration for theorists. Additionally, a half-integer spin quantum number would naturally produce exactly two possible orientations in a magnetic field — precisely the two beams that Stern and Gerlach had observed four years earlier.
On the advice of their mentor, Paul Ehrenfest, Uhlenbeck and Goudsmit wrote up the idea as a short note and submitted it for publication. It appeared in print on October 17, 1925. The paper included an acknowledgment of the model’s most obvious weakness: if the electron were a physically rotating sphere of known size, points on its surface would have to be moving faster than the speed of light. The authors flagged this openly, but pressed ahead regardless.
Early Criticism: Hendrik Lorentz and the Speed of Light Problem
The response from the broader physics community was cautious at best, and in some quarters openly skeptical.
Hendrik Lorentz, one of the most respected theoretical physicists in Europe at the time, examined the proposal carefully and concluded that it didn’t hold together. His calculation showed that for the model to be internally consistent, the electron would need to have a radius roughly comparable to the size of an atomic nucleus, many orders of magnitude larger than any experimental evidence suggested. Beyond the numbers, Hendrik Lorentz was blunt in his personal assessment. He told Uhlenbeck directly that such a reckless publication could only be attributed to the inexperience of young researchers who hadn’t yet developed the caution that serious physics demanded.
The criticism hit hard. Uhlenbeck seriously considered withdrawing the paper, and for a period, his confidence in the result wavered considerably. The faster-than-light problem wasn’t a minor technical quibble — it was a direct conflict with special relativity, one of the most firmly established frameworks in modern physics.
The mood shifted somewhat when a letter arrived from Werner Heisenberg. Rather than dismissing the work, Heisenberg congratulated Uhlenbeck and Goudsmit — not necessarily on having solved the problem, but on having the courage to put the idea into print at all. He raised several pointed questions and offered suggestions, but his tone was genuinely encouraging. For two physicists who had just been told their work was reckless, hearing from Heisenberg was no small thing.
Niels Bohr and the Acceptance of Electron Spin
By December 1925, the debate had grown prominent enough that Niels Bohr traveled to Leiden specifically to engage with it. On his way, he passed through Hamburg and stopped to consult both Pauli and Otto Stern, asking each of them directly for their assessment of the spin hypothesis.
When Bohr arrived in Leiden, he sat down with Ehrenfest and Einstein to work through the central difficulties. His main concern was a specific conceptual problem: an electron moving through the electric field of an atomic nucleus ought, in principle, to experience a magnetic field as a consequence of its own motion — a phenomenon known as spin-orbit coupling — and this interaction was supposed to account for the fine structure of spectral lines. But the mathematics didn’t come out correctly without something extra.
Einstein had already given this problem considerable thought and offered a resolution. From the perspective of the electron’s own rest frame — imagining, in other words, that you are traveling alongside the electron — the positively charged atomic nucleus appears to be moving in a closed orbit around the electron rather than the other way around. That orbiting positive charge generates a magnetic field at the electron’s location. The key was to perform the transformation between reference frames using the full machinery of special relativity rather than a classical approximation. When handled correctly, the result aligned with experimental observations.
With that obstacle cleared, Bohr became one of the most vocal advocates for the spin hypothesis. He encouraged Goudsmit and Uhlenbeck to develop their short note into a full research paper, which they subsequently did, with Bohr contributing a supportive commentary to the published version. Traveling onward to Göttingen and then Berlin, Bohr discussed spin with Heisenberg, Jordan, and eventually with Pauli himself, who, characteristically, referred to it as the “new Copenhagen heresy.” Despite his skepticism, Pauli would eventually come to accept it. Bohr’s enthusiasm by this point was such that in a letter to Ehrenfest, he described himself as “a preacher of the gospel of electron magnetism” — an unusually colorful expression for someone of his temperament.

Image credit: Peng, licensed under CC BY 3.0.
What Is Electron Spin? A Quantum Property Without a Classical Equivalent
The honest answer is that the question is more complicated than it first appears — and in certain respects, it remains so today.
The image of a literally spinning electron was always understood, at least by the more careful theorists, to be a useful analogy rather than a precise physical description. The faster-than-light objection raised by Hendrik Lorentz made clear that the classical picture of a rotating charged sphere couldn’t be taken at face value. Spin is not a physical rotation in the ordinary mechanical sense. It is a genuinely quantum mechanical property — one that has no direct counterpart in classical physics and cannot be fully visualized in everyday terms. It simply exists as an intrinsic characteristic of the electron, as fundamental as its mass or electric charge.
What spin did accomplish, however, was considerable. It resolved the anomalous Zeeman effect, explained the two-beam result of the Stern-Gerlach experiment, and provided the physical basis for Pauli’s exclusion principle — the rule governing how electrons arrange themselves in atoms, which underlies the entire structure of the periodic table and, by extension, the chemistry of everything. Spin also turned out not to be unique to electrons. Protons, neutrons, quarks, neutrinos, and a wide range of other elementary particles all carry spin. It is one of the most universal properties in all of physics.
The Stern-Gerlach experiment, originally designed to test a fairly specific prediction of early quantum theory, ended up doing something far more significant. It revealed that the internal architecture of matter was stranger and richer than anyone had anticipated. And the path from that puzzling two-beam result to a coherent theory of electron spin involved wrong guesses, sharp disagreements, one badly shaken young physicist, and eventually the collective effort of some of the greatest scientific minds of the 20th century.
References:
- Franklin, Allan; Perovic, Slobodan. “Experiment in Physics, Appendix 5”. In Zalta, Edward N. (ed.). The Stanford Encyclopedia of Philosophy
- Friedrich, B.; Herschbach, D. (2003). “Stern and Gerlach: How a Bad Cigar Helped Reorient Atomic Physics”
- Castelvecchi, D. (2022). “The Stern–Gerlach experiment at 100”. Nature Reviews Physics. 4 (3): 140–142.
- Reinisch, G. (1999). “Stern–Gerlach experiment as the pioneer—and probably the simplest—quantum entanglement test?”
